When a water is in balance, it is said to be neither corrosive nor scale-forming. In other words, it will not deposit a layer of calcium scale neither will it dissolve an existing layer of scale.
For most well run pools, the water will be in balance if the pH value is kept within the recommended range, but other factors should be taken into account which can affect the condition of the water. These are the total alkalinity, the calcium hardness, the TDS content and lastly, the temperature of the water. The concentration of chlorine or bromine do not appear in the Balanced Water Calculation.
The formula for determining whether he water is balanced was developed by Langelier in the 1930’s, hence the result after applying this is often called the Langelier Index or the Langelier Saturation Index or simply the Balanced WaterCalculation.
Why is the balance so important? Because if it is not correct, corrosion and erosion are possible.
There are 3 main causes of corrosion and erosion:
- Galvanic attack
- Aggressive water
- Low calcium hardness
Galvanic attack occurs when two or more dissimilar metals are in close proximity in a water environment (pool or spa ) which contains high levels of chemical salts or TDS. The presence of chlorides encourages the water to be more conductive. To prevent this the TDS can be reduced or the level of calcium hardness raised so that
a thin layer of scale is laid down to inhibit the metal’s efficiency as an electrode.
Lower levels of chlorides will prevent the water from acting as an electrolyte.
Low calcium hardness will often result in the loss of grout around the tiles, as the water tries to satisfy its need for calcium.
It is necessary therefore to maintain the TDS at sensible levels (ideally no more than 1000 mg/l above the feed water ) and yet maintain an adequate level of calcium hardness in the water (around 200 mg/l minimum ).
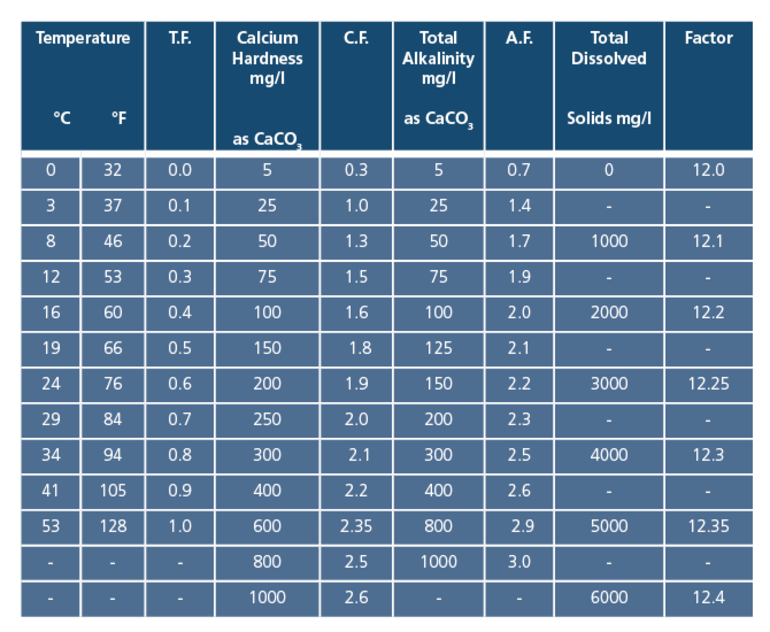
The formula for calculating the Langelier Index is as follows:
pH + Temperature factor + Alkalinity factor + Calcium Hardness factor - TDS factor
And is applied using the table below to obtain the factors from the actual test results for Total Alkalinity, Calcium Hardness and TDS.
In practical terms an Index Value in the region of zero to + 0.3 is considered satisfactory i.e. a low positive result, which indicates that the water can lay down a thin layer of protective scale.
Important note
A high Total Alkalinity is no compensation for a low Calcium Hardness. Each parameter should be within its recommended range.
Additional Water Balance Considerations
- In soft water areas where the constant addition of calcium is necessary to maintain a calcium level above the minimum, it could be advantageous to use calcium hypochlorite as the chlorine donor in order to obtain the calcium in addition to the chlorine from this product.
Also where the natural total alkalinity is low, the use of carbon dioxide gas for pH correction with calcium hypochlorite would be advantageous to produce an increase in the total alkalinity. - In hard water areas where it may be difficult to reduce total alkalinity and pH to the recommended range, the use of hydrochloric acid (muriatic acid) may be necessary and it may be appropriate to operate with a total alkalinity of around 140 – 150 mg/l.